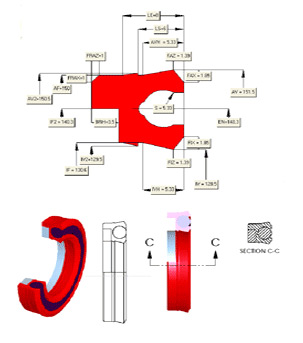
 When considering seal design for FDA seals, specifically for food contact, there are 5 factors that must be considered:
When considering seal design for FDA seals, specifically for food contact, there are 5 factors that must be considered:
1. Operating conditions
This might sound obvious, but a seal designer may focus on different concerns than you would with regard to the operating conditions. Too often a seal designer is asked to focus on one particular parameter and, upon closer review, it is the combination of two or three parameters that present the real challenge.
Here are the application details you should share with your seal designer:
• Type of movement(s)
• Operating pressure, normal speed, and temperature at this pressure
• Maximum pressure, and speed and temperature at this pressure
• Maximum speed
• Maximum temperature
2. Seal fitment and machine assembly
Many FDA seals have fitment issues that can be addressed at the initial design stage. If these are not addressed at this stage, these issues can present insurmountable obstacles at latter stages of the production process. Many seals require an open housing and will either be damaged or impossible to fit in a closed gland. The direction of insertion of the seal into the housing and shaft through the seal can also present real challenges.
We strongly recommend that you provide your seal manufacturer with a general assembly drawing as early as possible in the process to overcome or minimize this issue.
3. Housing and counter surface materials
FDA seals for the food industry use many housing, piston and shaft materials that present challenges to seal design. I have lost count of how many times I have had a design in mind only to be told, “Oh, by the way, the shaft is an exotic material. Is that a problem?”
Counter surface materials can drastically reduce the possible seal materials available to the designer and should be communicated to the design team ASAP. For example, an aluminum housing necessitates a different seal material than a stainless housing does.
4. Media being sealed
The media being sealed is especially important if there is either the possibility that the media will be changing in operation, or that the equipment being designed is to be suitable for a wide range of media. The media is an important factor driving the seal design and seal material selection. Sometimes it is necessary to have more than one sealing option available, depending upon the media.
5. Cleaning method
How the equipment will be cleaned in service can have a huge effect on seal design. It is not uncommon to discover that the best seal design and material for the application are totally unsuitable for the intended cleaning method. As soon as this is known, it should be communicated to the seal designer to ensure the application, media and cleaning method to be deployed are all considered together.
The best way to make the FDA seal design process go smoothly is to provide the information above to a seal designer at the very beginning of the process. Then the best recommendation can be given for a suitable seal that provides the longest possible life in service.
American High Performance Seals
www.ahpseals.com




Leave a Reply